Preventive Controls for Animal Food
go.ncsu.edu/readext?513009
en Español / em Português
El inglés es el idioma de control de esta página. En la medida en que haya algún conflicto entre la traducción al inglés y la traducción, el inglés prevalece.
Al hacer clic en el enlace de traducción se activa un servicio de traducción gratuito para convertir la página al español. Al igual que con cualquier traducción por Internet, la conversión no es sensible al contexto y puede que no traduzca el texto en su significado original. NC State Extension no garantiza la exactitud del texto traducido. Por favor, tenga en cuenta que algunas aplicaciones y/o servicios pueden no funcionar como se espera cuando se traducen.
Português
Inglês é o idioma de controle desta página. Na medida que haja algum conflito entre o texto original em Inglês e a tradução, o Inglês prevalece.
Ao clicar no link de tradução, um serviço gratuito de tradução será ativado para converter a página para o Português. Como em qualquer tradução pela internet, a conversão não é sensivel ao contexto e pode não ocorrer a tradução para o significado orginal. O serviço de Extensão da Carolina do Norte (NC State Extension) não garante a exatidão do texto traduzido. Por favor, observe que algumas funções ou serviços podem não funcionar como esperado após a tradução.
English
English is the controlling language of this page. To the extent there is any conflict between the English text and the translation, English controls.
Clicking on the translation link activates a free translation service to convert the page to Spanish. As with any Internet translation, the conversion is not context-sensitive and may not translate the text to its original meaning. NC State Extension does not guarantee the accuracy of the translated text. Please note that some applications and/or services may not function as expected when translated.
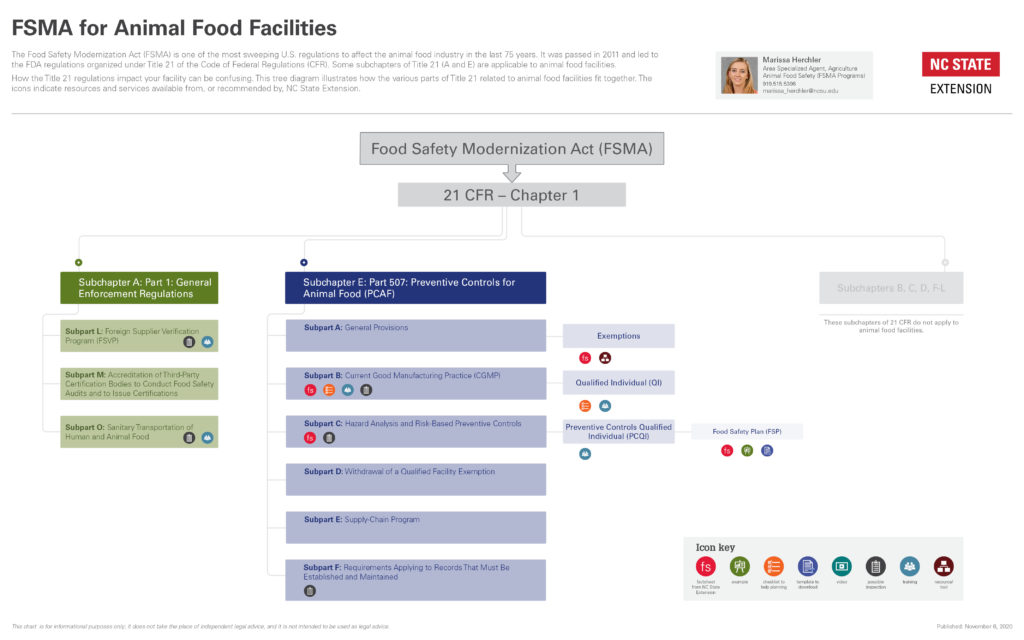
Collapse ▲The Food Safety Modernization Act (FSMA) of 2011 can be difficult to implement because it’s so far-reaching. Our pages explain some of the components of the rules that impact food for animals.
This page provides information about what the Preventive Controls for Animal Food (PCAF) rule is and provides an outline of other pages that are helpful when implementing this rule in your facility.
The PCAF rule is part of the regulations introduced by the FDA after passage of FSMA in 2011.
For an overview of how PCAF fits in with FDA regulations related to animal food, see our diagrams below.
Where should I begin?
Each page linked below includes background information, key messages and available resources about a section of the Preventive Controls for Animal Food rule.
We recommend starting by reading through these pages to become more familiar with the PCAF rule and how it might impact your establishment’s food safety journey. Then contact Marissa Herchler Cohen with any questions you may have.
Each facility’s food safety journey is as unique as their business and the people who work there. Marissa works with facilities to help determine how they should comply with the rules and to help get them started.
The PCAF rule can be overwhelming for many. This page provides a general overview of the PCAF rule and the subparts of the rule with resources that help increase understanding of the components of the rule and its subparts. These resources are important for understanding how they fit together to improve food safety standards.
Some of the terms used in the PCAF rule are familiar, while some may mean something completely different from what you might think. Use this page to navigate through some of the terminology used in the rule that might not be defined in the definitions listed in 21 CFR 507.3.
Some facilities may be required to comply with none or only certain parts of the rules. Use these resources to help determine where your facility falls.
PCAF: Current Good Manufacturing Practice
Current Good Manufacturing Practice (CGMP) are baseline food safety standards found in Subpart B of the PCAF rule. Explore this page to learn more about the CGMP requirements and resources NC State Extension has created.
Training requirements are new to the industry and are a significant part of complying with the FSMA rules. Know what training is needed to be in compliance with the rules and where to find training courses.
Food safety plans (FSPs) are also new requirements for many facilities. Find out what an FSP should (& shouldn’t) contain and see example plans as they become available.
We’ve put together a list of documents and organizations that might be helpful as you develop a food safety plan, in addition to the resources our team offers. Visit this page to learn more about these organizations and resources.
For additional information contact
Marissa Herchler Cohen
NC State University
Cooperative Extension Service
Prestage Department of Poultry Science
234 D Scott Hall
Raleigh, NC 27695-7608
E-mail: marissa_cohen@ncsu.edu
Phone: +1 919.515.5396